04 Jan Predicting the effects of drugs that modulate calcium transients
Disopyramide
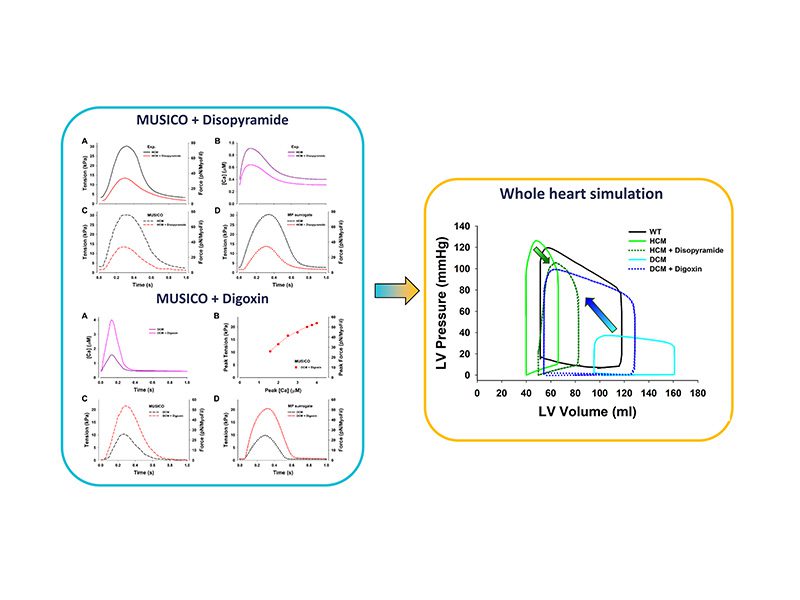
Disopyramide, classified as a type I antiarrhythmic (i.e., a sodium channel blocker) is a potent negative inotrope. It has been used to decrease left ventricular outflow tract obstruction in obstructive HCM since first reports in the early 1980’s. Until recently it was not known whether the drug had direct effects on Excitation-Contraction Coupling or on the actin-myosin interaction. Recent work by Coppini et al. (2019) on human HCM cardiomyocytes and trabeculae has shown that disopyramide inhibits multiple ion channels, leading to lower Calcium transients and twitch force with no direct inhibitory effects on myofilament function. In addition, disopyramide does not exert proarrhythmic effects in vitro, rather it shortens action potentials showing potential for reduced arrhythmic propensity. The observation that disopyramide is a negative inotrope that does not interfere with sarcomere protein function makes this drug likely compatible with the new class of allosteric myosin modulators (e.g., Mavacamten) currently under successful clinical investigation for use in HCM. MUSICO simulations predicted decrease on the peak tension by ~55% and decrease in resting tension by ~50% in the presence of 5 µmol/l of disopyramide, matching the observations of Coppini et al. (2019).
Digoxin
Cardioactive glycosides (e.g., Digoxin) have been important in treating congestive heart failure for more than 200 years, in large part because of a positive inotropic effect. The traditional view is that direct Na+–K+-ATPase inhibition leads to elevation of [Na+]i with consequent gain of cellular and sarcoplasmic reticulum (SR) calcium concentration as a result of shifts mediated via the Na+–Ca2+ exchanger. That is, with higher [Na+]i, calcium extrusion via NCX is reduced and calcium influx via NCX may be enhanced. Thus, there can be higher diastolic [Ca2+]i (closer to threshold for contractile activation), higher SR calcium content (causing greater SR calcium release during contraction) and even greater calcium influx via NCX during the action potential. All these mechanisms lead to increased calcium transient amplitude and peak twitch force. Studies with aequorin injected preparations of human cardiac muscle showed that cardiac glycosides increase twitch force by increasing the amplitude of the calcium transient without affecting the time course of the calcium transient and that the increase in tension is fully accounted for the increase in systolic free calcium. The increase in intracellular calcium concentration increases heart wall tension, and therefore increases systolic pressure and ejection fraction. The MUSICO simulations predicted dose dependent increase on the peak tension up to twofold. The peak of calcium concentration during muscle contractions corresponds to different dosages of digoxin taken from the literature.
The MUSICO simulations for both drugs are approximately replicated by MP surrogate model and used in left ventricle heart simulations using Finite Element solver to quantitatively assess the effect of drugs on cardiac output including decrease in both systolic and diastolic pressures, and the ejection fraction. Simulations showed that both drugs improve performance of hypertrophic (HCM) and dilated (DCM) cardiomyopathy hearts. The principal effect of Disopyramide is significant decrease in magnitude of calcium transient and the activation patterns that is reflected in decrease in peak tension and shift toward higher volumes. In contrast, Digoxin showed large increase (several fold) of the calcium transient magnitude that causes large increase in ventricular peak pressures which shifts the P-V loop of DCM left ventricle towards the P-V loop observed in healthy hearts.




No Comments