02 Jan Predicting the effects of drugs that affect changes in kinetic parameters
Mavacamten
Mavacamten (MYK-461) was recently approved by FDA as an allosteric inhibitor of sarcomeric myosins since it had been successfully used in clinical trials for treatment of obstructive HCM. Mavacamten negative inotropic action is likely mediated by the shift of detached motor heads towards auto inhibited SRX state. The drug reduces peak twitch tension of intact human ventricular muscle without major changes in the time course of the twitch. The impact of Mavacamten on cardiomyocyte electrophysiology and calcium handling is still under investigation, but the drug is able to reverse the adverse remodeling of cardiomyocyte excitation-contraction coupling observed in mouse models of HCM.
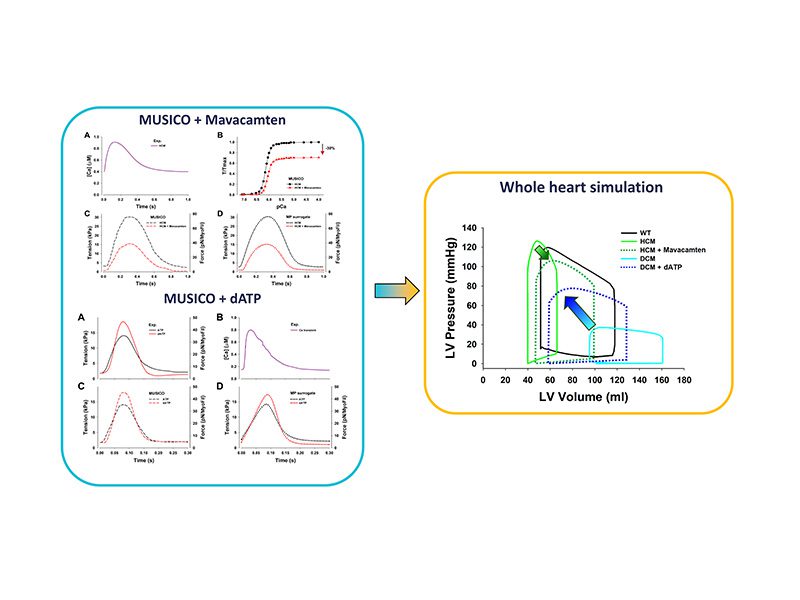
The effect of increased tension in HCM can be attenuated by mavacamten action on the level of crossbridge cycle, specifically by modulation of the state transition rates between the parked state and disordered myosin detached states, capable of binding to actin. The model parameters for the change of these rates are obtained from MUSICO fits of force in HCM cardiac muscles in absence or in presence of 1 µm mavacamten. Using the same calcium transient as observed in HCM, and crossbridge parameters without and with mavacamten showed significant decrease in tension, ~30% for steady state force development at high calcium concentrations and about 50% in twitch contractions. Furthermore, simulations also showed significant decrease in resting tension. These changes are similar to the tension responses as predicted by disopyramide but the mechanisms of the action of these two distinctive drugs are fundamentally different. These data show flexibility of MUSICO platform to quantitatively predict distinctively different mechanisms of drugs for the treatment of HCM patients.
2-deoxy adenosine triphosphate (dATP)
The molecule 2-deoxy adenosine triphosphate (dATP) can replace ATP as the energy source for the motor protein myosin contraction in striated muscle. dATP allosterically enhances myosin crossbridge binding to actin (and cycling kinetic) such that small amounts of dATP are potent. Increasing the cardiomyocyte level of dATP from the typical <0.1% of the ATP pool to just 1% is enough to significantly increase contraction. In rodent and pig models of myocardial infarct and heart failure dATP improves left ventricular function, including developed systolic pressure, the rate of pressure development and the rate of pressure decline at the end of systole. It has been also demonstrated that dATP increases contractility of cardiac tissue from heart failure patients. Thus, approaches to increase cardiomyocytes dATP constitute an exciting and novel therapy with the potential to treat heart failure.
The significant increase in cardiac muscle contractility could be beneficial for DCM cardiomyopathies. Using MUSICO simulations, we quantitatively assessed the effect of dATP as the energy source on twitch contractions in mice trabeculae. Differences in estimated MUSICO parameters are only in myosin-actin binding and detachment rates while keeping the equilibrium binding rate constant resulting in higher force generation and faster rise and relaxation times.
The MUSICO simulations for both drugs are approximately replicated by MP surrogate model and used in left ventricle heart simulations using Finite Element solver to quantitatively assess the effect of drug on cardiac output including decrease in both systolic and diastolic pressures, and the ejection fraction. Simulations showed that both drugs improve performance of hypertrophic (HCM) and dilated (DCM) cardiomyopathy hearts. The principal effect of Mavacamten is significant decrease in available myosin heads in acto-myosin cycle which is reflected in decrease in peak tension and shift toward higher volumes. In contrast, dATP showed large increase in muscle contractility that causes large increase in ventricular peak pressures which shifts the P-V loop of DCM left ventricle towards the P-V loop observed in healthy hearts.




No Comments